Use of Chloroplasts for Oxygen Production in Cell Cultures
In varied medical and biotechnological settings, an abundance of cellular oxygen is often required to facilitate proper cellular function, promote cell growth, or prevent irreversible tissue death. As such, there exists an impetus for the development of cheap and efficient technologies that can produce oxygen at the cellular level for use on the cellular level. Such a technology could be used to enhance cell cultures with integrated oxygen production, to grow full organs or tissues in vitro, to vascularize and regenerate ischemic tissues, to accelerate wound healing, to be used in biotechnology production plants, to enhance human performance and physical ability, to produce oxygen in hypoxic environments, and for myriad other applications in which a surplus of cellular oxygen would be beneficial.
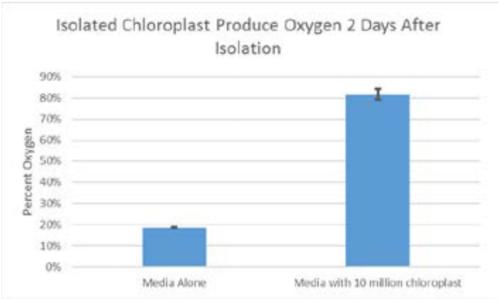
These two novel methods of culturing chloroplasts, both extracellularly and intracellularly, within a cellular media can easily produce large quantities of oxygen for cell cultures and tissues as well as grant human cells the ability to photosynthesize.
Competing approaches focus primarily on the extraction and isolation of a vast array of biological materials as well as chloroplasts, often relying on mechanical support systems for adherence of the isolated biological material. Some approaches instead explore the use of chloroplasts for energy production when introduced to certain metals.
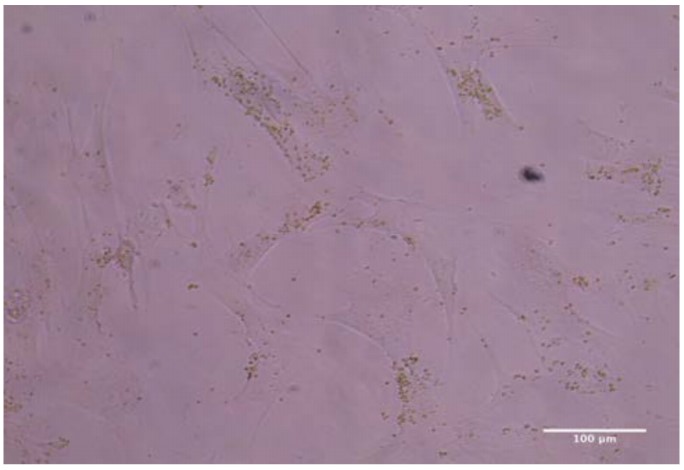
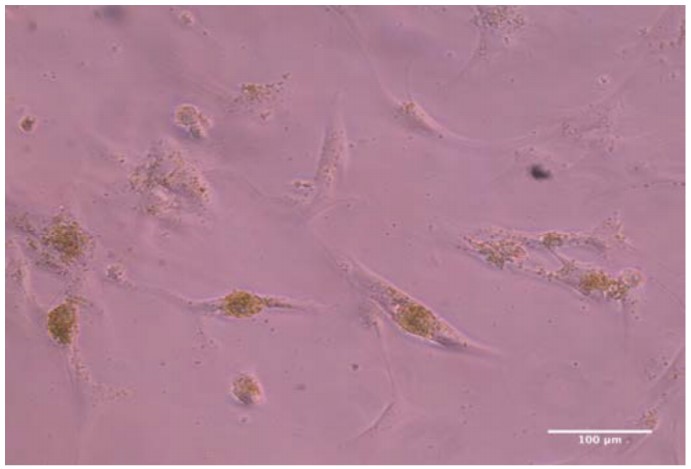
This approach is the first to detail the isolation of chloroplasts specifically from spinach leaves. It is also novel in its demonstration of the ability to integrate isolated chloroplasts within human cells. This intracellular incorporation of chloroplasts should allow for greatly improved oxygen production to be used in possible cell therapies or biotechnology production plants.

Key Features/Benefits
Not only does this approach present a novel method for incorporation of chloroplasts amongst human cells in culture, it also introduces a method for the integration of functional chloroplasts within human cells themselves, effectively creating chloroplast-human cell symbionts with significant lifespans. These two methods represent potential mechanisms for the delivery of oxygen to human cellular tissues and cultures alike. Additionally, this process for creation of chloroplast-human cell symbionts opens the door for countless potential applications in medicine and beyond. Lastly, this approach also details a new process for the isolation of chloroplasts from spinach leaves.