Research

Research is a major part of the mission and day-to-day life of the Department of Chemistry and Biochemistry. Our faculty and students conduct groundbreaking research that has practical applications in everyday life–from the heath care and medications we receive to the energy we use to make our world go, and beyond.
Our research laboratories are housed in the Life Sciences and Bioengineering Center at Gateway Park, a state-of-the art, major research facility in which bays are organized by research focus rather than by department and researchers from the full range of life science disciplines freely exchange ideas.
Learn about our research interests of faculty members in the WPI Department of Chemistry & Biochemistry:
José M. Argüello
In his busy lab, José Argüello and his group are studying the biochemical ballet by which cells transport metal micronutrients, such as copper, zinc, cobalt, and iron, across their membranes and to the sites where they play such fundamental roles as helping blood transport oxygen or plants fix nitrogen from the air. Because of the importance of these basic biological functions, a better understanding of the mechanisms of heavy metal transport has implications for the treatment of a host of diseases like tuberculosis, for human and animal nutrition, and for the bioremediation of heavy metal pollution.
He has received multiple research grants from the National Science Foundation (NSF) and the National Institutes of Health (NIH), including an NIH Research Development Award for Minority Faculty in 1995 and a $1.3 million award in 2016 for a systematic study of copper in the bacteria Pseudomonas aeruginosa, a leading cause of hospital-associated infections. He was recently elected Fellow of the American Association for the Advancement of Science (AAAS), the world’s largest general scientific society, “for distinguished research discoveries elucidating the mechanisms underlying metal ion transport and the role of bacterial metal transporters in agriculture and infectious disease.” Research interests include: Mechanisms of micronutrient metal transport in biological systems; membrane protein chemistry; role of micronutrient metals in pathogenic virulence and endocytic symbiosis; role of bacterial metal transport proteins in tuberculosis and in plant nitrogen fixation.
Learn more about Professor Argüello and the Argüello research group.
Shawn C. Burdette
Prof. Burdette investigates multidisciplinary research at the interface of metals, biology and photochemistry. Most of the projects in the Burdette group begin with the synthesis of chemical tools that are designed to execute a particular task such as bind and release a metal of interest. The new chemical tools are then characterized spectroscopically and applied to answering a question in a related field like biology either within the group or in collaboration with other groups. Research interests include: Bioorganic zinc chemistry; metals in neurotransmission; metal ion signaling in biology; metal in homeostasis; metal ion chelators: binding and selectivity; organic photochemistry/photochemical tools; small-molecule and polymer-based fluorescent sensors; new fluorophores and chromophores; ligand synthesis; living polymerization methods.
Learn more about Professor Burdette and the Burdette research group.
Bruce E. Bursten
Bruce Bursten conducts research on the correlation of theoretical and experimental electronic structural data with the bonding and reactivity patterns of metal-containing molecules. Systems currently under investigation include the bonding in compounds of the actinide elements (such as U and Pu), photochemical behavior of dinuclear organometallic complexes, and the nature of metal-metal multiple bonds. His group primarily uses density functional theory calculations as a probe of electronic structure.
Prof. Bursten has published more than 160 research papers and coauthors a leading chemistry textbook, currently in its 15th edition. He was elected President of the American Chemical Society (ACS) for 2008 and Chair of the AAAS Section on Chemistry for 2015. Among his honors, he has been named a Camille and Henry Dreyfus Foundation Teacher-Scholar, a Sloan Fellow, and Fellows of the AAAS and the ACS. He has received the Catalyst Award from the American Chemistry Council, the Spiers Medal of the Royal Society of Chemistry, the ACS Morley Medal, and the ACS National Award for Distinguished Service in the Advancement of Inorganic Chemistry.
Learn more about Professor Bursten.
Robert E. Dempski
Prof. Dempski’s research focuses on understanding the mechanism of biomedically essential, but understudied membrane proteins. This includes the human zinc ZIP4 transporter which has been implicated in the initiation and progression and solid tissue tumors such as pancreatic cancer as well as the light activated channelrhodopsin-2 ion channel, a central tool in the emerging field of optogenetics.
Learn more about Professor Dempski and the Dempski research group.
Arne Gericke
Prof. Gericke is interested in studying signal transduction pathways and investigating how lipids mediate protein functions – in particular, the PI 3-kinase signaling pathway, which is one of the major pathways involved in the development of cancer. Prof. Scarlata and Prof. Gericke's group uses sophisticated spectroscopic, kinetic and thermodynamic methods to study lipid/protein interaction in cells and model systems. Research interests include: Membrane biophysics; lipid-mediated protein functions (phosphoinositides, sphingolipids); membrane trafficking; vibrational spectroscopic imaging of tissue.
Learn more about Professor Gericke and the Gericke research group.
Ronald L. Grimm
The Grimmgroup is interested in understanding what makes a particular material efficient at converting sunlight to electrical or chemical energy? Conversely, what makes a material a poor energy converter? Using a range of spectroscopic techniques, the group seeks an atom- and bond-level understanding of the properties of non-traditional materials for energy conversion and storage.
Supramolecular chemistry is the design of molecular materials and structures that exhibit useful properties resulting from the collective interaction of different atomic components. The Grimmgroup is motivated by quantifying and controlling the bulk and surface properties of solar energy conversion materials, energy storage materials, inorganic-organic interfaces, and barriers against chemical warfare agents.
Learn more about Professor Grimm and the Grimmgroup.
Destin Heilman
Dr. Heilman and his lab is working to discover how a virus can destroy only cancerous cells directly with the hope of developing new cancer therapies. He spoke to Nature.com in a video explaining how viruses function and how the machinery of a virus could be hijacked to cure diseases. Research interests include: Animal virus protein; apoptosis; cancer cell selectivity; subcellular localization; functional domain mutagenesis.
Learn more about Professor Heilman and the Heilman research group.
George A. Kaminski
Prof. Kaminski is a computational physical chemist who models small molecule/protein interactions. Toxic proteins are at the root of many diseases, so pharmaceutical companies look for compounds called ligands that will bind tightly to these proteins and deactivate them. He has developed powerful new computational tools that will make the search faster and more precise.
The tools use principles of physics to predict protein-ligand binding based on the chemistry and structure of target compounds. Research interests include: Computational chemistry/molecular modeling; protein simulations; force field development; Monte Carlo simulations.
Learn more about Professor Kaminski and the Kaminski research group.
Christopher R. Lambert
Antimicrobial surfaces for device implantation; neuroprosthetics for regenerating nerves and enabling devices; passive cell sorting for blood purification; bioreactor development for tissue growth, manufacturing therapeutics, and creating biofuels; organic photovoltaics; acetylcholinesterase inhibition sensing; blood analytes sensing.
Learn more about Professor Lambert.
John C. MacDonald
Prof. MacDonald focuses on understanding molecular interaction and self-assembly in solution and on surfaces in order to control molecular crystallization and assembly of complex multicomponent systems. His group utilizes synthetic, physical-organic, and supramolecular approaches to generate and study 3-D coordination polymers—called metal-organic frameworks, or MOFs—featuring a range of architectures with high internal surface areas and void volumes. These porous MOF solids serve as host materials that actively absorb and store organic guests. Research interests include: Surface chemistry: molecular thin films; materials chemistry: porous solides (environmental remediation); solid-state organic chemistry: crystal engineering, polymorphism, chiral separations.
Learn more about Professor MacDonald.
Anita E. Mattson
Prof. Mattson is an organic chemist with a strong medicinal chemistry component in her research. Her research is directed at the synthesis of complex molecules that have pharmaceutical value or promise. Her studies looks at the preparation of important synthetic building blocks using new transformations catalyzed by small organic molecules. Of particular interest is the development of reactions catalyzed by organic molecules through non-covalent interactions. Her group applies these new synthetic methods towards the synthesis of naturally occurring bioactive targets.
She was recently was awarded a grant from the National Science Foundation (NSF) entitled "Chiral silanediols in anion-binding catalysis.” Under this grant, Dr. Mattson will design and implement studies related to chiral silanediols in anion-binding catalysis and will oversee two graduate students in the syntheses of chiral silanediols and the development of innovative enantioselective methodologies reliant on silanediol anion-binding catalysis. Dr. Mattson will also oversee mechanistic studies probing the noncovalent interactions of silanediol-catalyzed anion-binding processes.
Learn more about Professor Mattson.
Carissa Perez-Olsen
Prof. Perez-Olsen studies the maintenance of cell membrane composition in C. Elegans and how the lipidome is affected by age and/or disease state. In addition, she is interested in identifying genes that are important for regulating membrane maintenance under normal and stressed conditions. She uses advanced mass spectrometry to study the lipid composition of cell membranes. Her research aims to understand the linkage between biomembrane maintenance and diseases like cancer.
Learn more about Professor Olsen and the Olsen research group.
Suzanne F. Scarlata
Prof. Scarlata’s group explores how cells communicate with the outside world. This communication system can involve a group of proteins called “G-proteins” that can cause cells to move, divide and change structure. Using biophysical methods, the Scarlata group can watch how cells respond to different agents and try to control their responses.
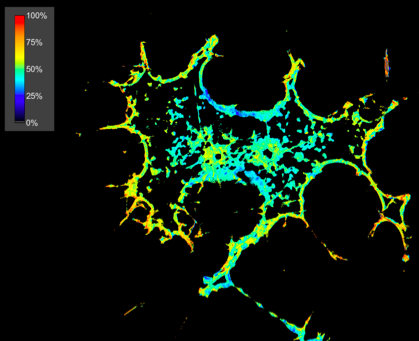 |
Suzanne Scarlata’s lab uses advanced fluorescence microscopy techniques to study membrane signaling events involving G-protein coupled receptors. FRET images of PC12 cells over-expressing eCFP-Gqα and eCYP-Phospholipase Cβ1. The high FRET intensity at the membrane (yellow/red color) indicates that Gqα/PLCβ1β dimer formation at the plasma membrane. |
The Scarlata lab employs sophisticated spectroscopic, kinetic and thermodynamic methods to study lipid/protein interaction in cells and model systems. Her own research, funded with an R01 award from the National Institute of General Medical Sciences (NIGMS), focuses on guanine nucleotide-binding proteins’—or G proteins’—role in transmitting signals to various stimuli in the body, particularly in heart muscle cells and neurons.
She is a recent president of the Biophysical Society, with over 9,000 global members. Her goals in the new role include efforts to establish a more stable U.S. research funding system, spreading the word about the biochemical and physical science realm of WPI, and forging more community collaborations with groups like UMass Medical School. Previously, she was the chair of the societies’ Committee for Professional Opportunities for Women. Prof. Scarlata has pioneered the adoption of the society's gender equity policy that made a 'commitment to inclusivity and diversity and not at the expense of scientific excellence'.
Rong R. Wang
The Wang group has expertise in bioconjugate chemistry, molecular imaging, nano/micro-fabrication, cell biology and material engineering. Research focuses on developing novel methods, materials and devices for detection and intervention of bioprocesses.
Students receive training in biochemistry, biophysics, analytical chemistry, surface chemistry, composite materials and cell biology through the following ongoing research projects: Examination of structure-function relationship for collagen in native tissues., Development of biocomposite materials as tissue engineering scaffolds, Salivary sensor for label-free detection of periodontal causative oral bacteria. IR-responsive polymer in sensor application, and others.