For researchers, the path to understanding aortic heart valve disease is littered with clues.
Chronic high blood pressure, or hypertension, is a risk factor. So is inflammation. And then there is lipoprotein (a), also known as Lp(a), a sticky cholesterol-carrying particle that circulates in the blood.
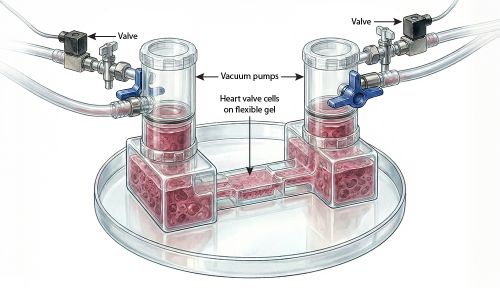
Kristen Billiar, a professor in the Department of Biomedical Engineering, will try to determine what turns those risk factors into disease as part of a $15 million multicenter initiative that is funded by the American Heart Association and focused on early detection and prevention of heart valve disorders.
Billiar has been awarded $1,278,213 for a four-year project aimed at determining the connection between circulating Lp(a), the biomechanical forces at work in the aortic valve, and development of calcific aortic valve disease (CAVD). CAVD is the most common heart valve disease, causing an estimated 248,256 deaths in the United States in 2019. The disease thickens, calcifies, and narrows the thin, flexible flaps of tissue that open and close in valves as blood moves through a beating heart.
Billiar’s project is one of three taking place under the Center for Aortic Valve Disease Prediction and Integrated Research at the University of Pittsburgh. Cynthia St. Hilaire, associate professor of medicine at the University of Pittsburgh, is the center director and also will lead a project with collaborators at Creighton University. A third team will be led by Satoshi Okawa, University of Pittsburgh assistant professor of medicine. Together, the three teams will focus on early detection, disease progression, and treatment of aortic stenosis, which is a narrowing of the heart valve that allows oxygen-rich blood to flow out to the body.
“The project at WPI will focus on biomechanics, but we will also collaborate with researchers at other institutions who are concentrating on cell biology and using artificial intelligence (AI) to examine genetic risk factors,” Billiar says. “The goal is to identify targets for potential therapies to treat heart valve disorders.”