WPI researchers have used a form of artificial intelligence (AI) to analyze anatomical changes in the brain and predict Alzheimer’s disease with nearly 93% accuracy.
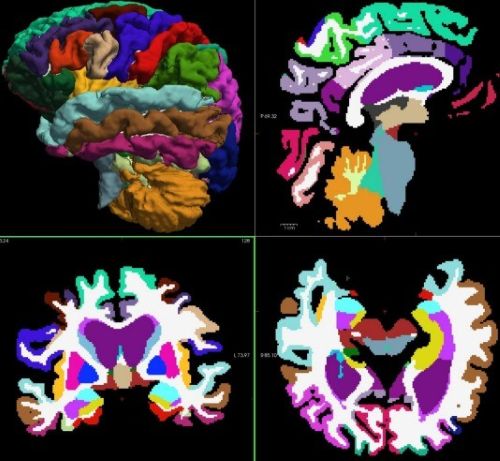
Segmented and labeled images of a normal brain
Their research, published in the journal Neuroscience, also revealed that the anatomical changes, involving loss of brain volume, differ by age and sex.
“Early diagnosis of Alzheimer’s disease can be difficult because symptoms can be mistaken for normal aging,” says Benjamin Nephew, assistant research professor in the Department of Biology and Biotechnology. “We found that machine-learning technologies, however, can analyze large amounts of data from scans to identify subtle changes and accurately predict Alzheimer’s disease and related cognitive states. This advance has informed Alzheimer’s disease research and may lead to methods that could allow doctors to diagnose and treat the disease earlier and more effectively.”
Alzheimer’s disease is a neurodegenerative disorder that impairs mental functions and ultimately leads to death. An estimated 6.9 million Americans age 65 and older are living with Alzheimer’s disease.
Healthy brains contain billions of neurons, the cells that process and transmit signals needed for thought, movement, and other bodily functions. Alzheimer’s disease injures neurons, leading to cell death and loss of brain tissue and associated cognitive functions.
Nephew, PhD student Senbao Lu, and Bhaavin Jogeshwar, MS ’24, conducted their research with MRI scans of brains from the Alzheimer’s Disease Neuroimaging Initiative, a multicenter project that built a library of brain scans from people age 69 to 84. The scans depict the brains of people with normal mental functioning, mild cognitive impairment, and Alzheimer’s disease.
Analyzing data-rich MRI images can require substantial computing power and time. To focus their investigation, the WPI researchers first used machine learning to analyze 815 MRI scans for volume measurements in 95 brain regions. Then they deployed an algorithm to make predictions based upon differences in the measurements between healthy individuals and those with mild cognitive impairment or Alzheimer’s disease.
Results showed that the method was 92.87% accurate in detecting Alzheimer’s disease among normal brains and brains of people with mild cognitive impairment.







