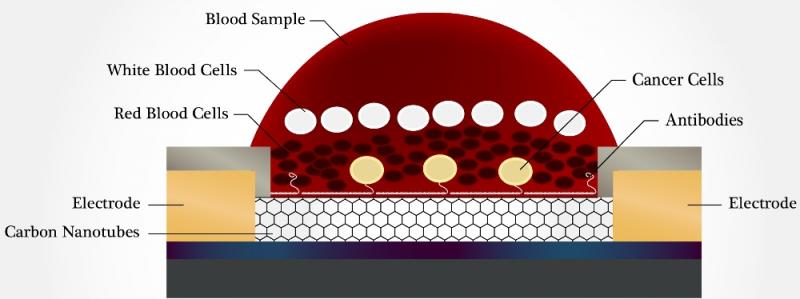
Panchapakesan said the chip developed by his team has additional advantages over other liquid biopsy devices, most of which use microfluidics to capture cancer cells. In addition to being able to capture circulating tumor cells far more efficiently than microfluidic chips (in which cells must latch onto anchored antibodies as they pass by in a stream of moving liquid), the WPI device is also highly effective in separating cancer cells from the other cells and material in the blood through differential settling.
While the initial tests with the chip have focused on breast cancer, Panchapakesan says the device could be set up to detect a wide range of tumor types, and plans are already in the works for development of an advanced device as well as testing for other cancer types, including lung and pancreas cancer. He says he envisions a day when a device like his could be employed not only for regular follow ups for patients who have had cancer, but in routine cancer screening.
“Imagine going to the doctor for your yearly physical,” he said. “You have blood drawn and that one blood sample can be tested for a comprehensive array of cancer cell markers. Cancers would be caught at their earliest stage and other stages of development, and doctors would have the necessary protein or genetic information from these captured cells to customize your treatment based on the specific markers for your cancer. This would really be a way to put your health in your own hands.”
“White blood cells, in particular, are a problem, because they are quite numerous in blood and they can be mistaken for cancer cells,” he said. “Our device uses what is called a passive leukocyte depletion strategy. Because of density differences, the cancer cells tend to settle to the bottom of the wells (and this only happens in a narrow window), where they encounter the antibodies. The remainder of the blood contents stays at the top of the wells and can simply be washed away.”