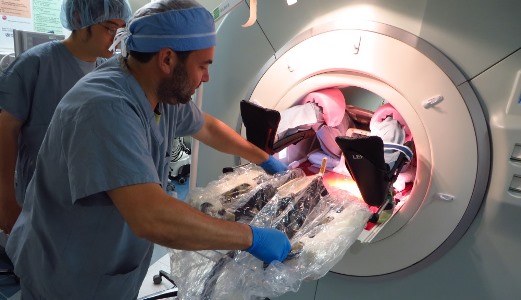
Some people design robots that can assemble circuit boards or vacuum your floor. Gregory Fischer, PhD (above), professor of mechanical engineering and robotics engineering and director of WPI’s Automation and Interventional Medicine (AIM) Laboratory (aimlab.wpi.edu), has other things in mind: He wants his robots to take biopsies from cancer patients more accurately and efficiently, and perform brain surgery more safely and effectively.
And he is tantalizingly close to achieving both milestones.
A Robotics Revolution in the AIM Lab
Fischer has made his mark by developing robots that can work inside MRI scanners, enabling surgeons to operate guided by real-time medical images. One robot, which he spent years developing in conjunction with colleagues at Johns Hopkins University, is in clinical trials at Brigham and Women’s Hospital (BWH) in Boston, where it is being used to help take biopsies from prostate cancer patients. (The trials are part of a program led by Clare Tempany, MD, a professor of radiology at Harvard Medical School and chair of research radiology at BWH, and supported by the National Institutes of Health’s National Center for Image Guided Therapy and with a Bioengineering Research Partnership grant from the National Cancer Institute.)
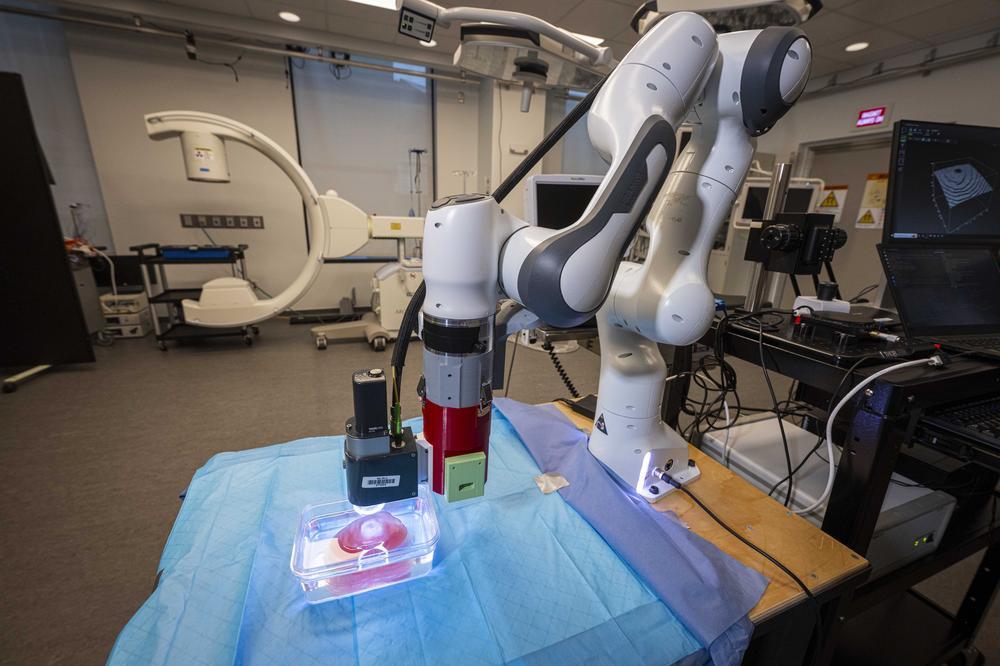
Another robot will blast brain tumors using high-powered, MRI-guided, robotically manipulated ultrasound. Now a fully functional prototype, that robot, supported by an NIH Academic-Industry Partnership grant and developed in collaboration with the University of Massachusetts Medical School, Albany Medical Center, and Acoustic MedSystems, could begin human trials in a few years.